Seeing Cancer Clearly
How Imaging Works and Why Your Oncologist Chooses One Scan Over Another
Editor’s note:
This piece comes from questions I hear every week in clinic. Imaging is an important part of cancer care, but the reasoning behind different scans is rarely explained. My hope is that this helps make those decisions feel clearer and more collaborative.
—Dan
Imaging is part of almost every cancer conversation. At some point, we start talking about scans and why one might make more sense than another. The goal is usually the same: to get a better understanding of what’s going on with the cancer right now and to help decide how to move forward.
These decisions shouldn’t be automatic. When I’m sitting with a patient and we’re talking about imaging, we’re trying to be clear about the question in front of us and how best to answer it. “Scanxiety” is real and being thoughtful about when and why we order these tests is part of good care.
Concerns about radiation exposure over time, prior experiences with contrast dye, or discomfort with MRI related to claustrophobia often shape the plan. Patient input helps guide which test fits best and helps ensure they’re not just along for the ride.
The scan is a tool. The person is the focus.
Once the question is clear, the next step is choosing the imaging that is most likely to give us useful information.
What imaging can and cannot show
No imaging test can see individual cancer cells. Scans do not detect microscopic disease or tiny clusters of cancer cells circulating in the bloodstream. All current imaging operates at a scale where disease has to reach a certain size or level of activity to be visible.
What imaging can show are patterns that are large enough to influence decisions. Changes in tumor size. Differences in density or signal. Areas that appear more metabolically active. Those findings help us track disease over time and evaluate response to treatment, even though they are always indirect measures of what’s happening biologically.
That limitation applies to every imaging modality and explains why scans are helpful, but never definitive on their own.
CT scans: anatomic assessment and longitudinal tracking
CT scans, sometimes called CAT scans, are the most commonly used imaging test in oncology. They rely on X-rays to create detailed cross-sectional images and are particularly good at showing anatomy. Tumor size, shape, and location are where CT performs best.
CT is often the backbone of cancer imaging at diagnosis, during treatment, and in surveillance afterward. It allows for consistent measurements over time, which is critical when we’re trying to determine whether disease is responding, stable, or progressing. CT scans are fast and widely available, and the short scan time can be an advantage for patients who are uncomfortable or anxious.
CT scans do involve ionizing radiation, though typically less than PET scans. Many CT studies also use contrast, either oral, intravenous, or both. Iodine-based contrast improves visualization of organs, blood vessels, and lymph nodes, which often makes interpretation more reliable. That benefit has to be balanced against prior reactions, kidney function, and patient tolerance.
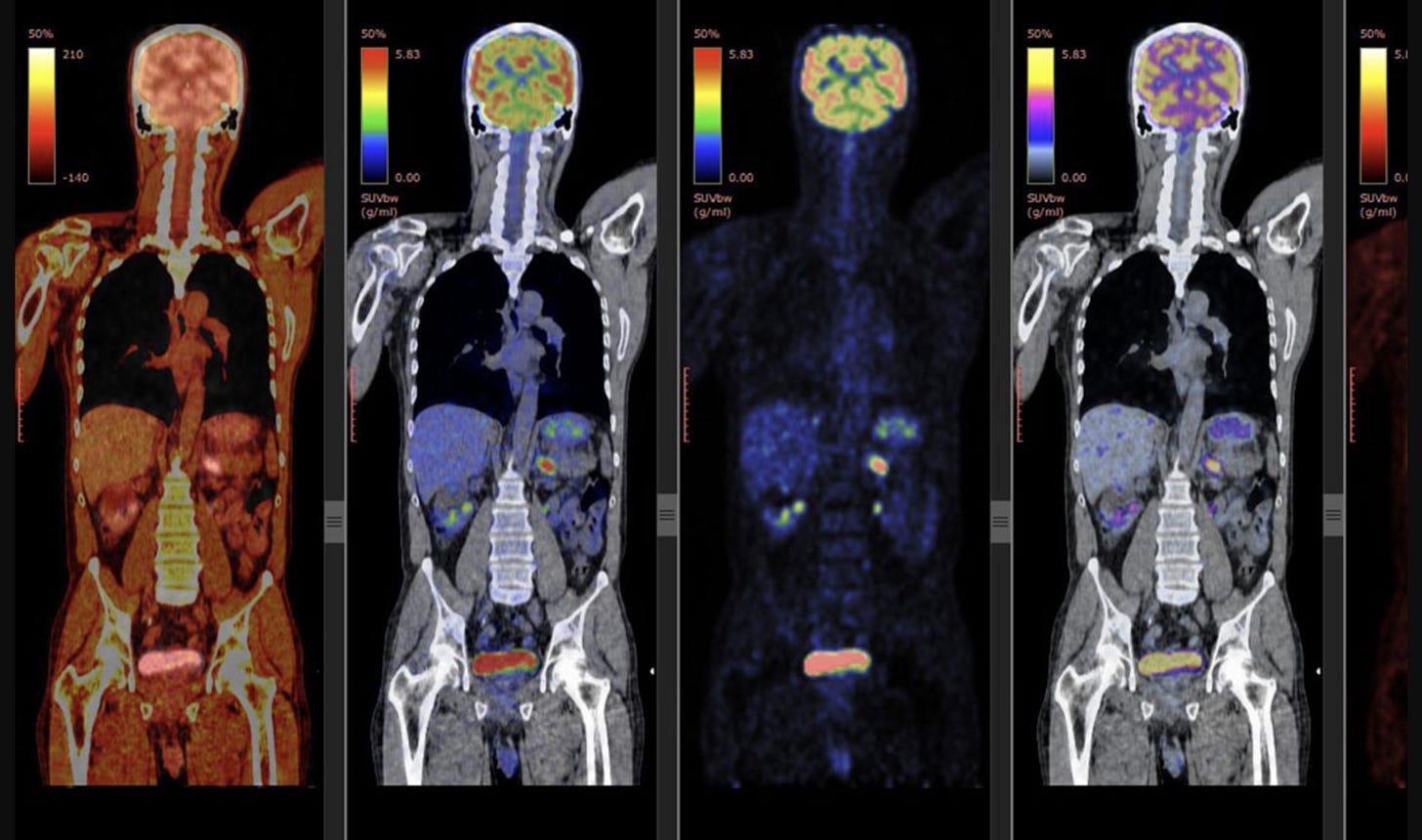
PET scans: metabolic activity and biologic signal
PET scans provide a different layer of information. Rather than focusing on structure, they assess metabolic activity. We often describe this activity as “hot” or “cold.”
Most PET scans in oncology use a glucose-based radiotracer called FDG. Because many cancer cells have altered metabolism and increased glucose uptake, areas of active disease often appear “hot” as regions of increased signal activity. This can allow PET scans to identify disease that may not yet be causing obvious anatomic changes on CT. This can be quantified as well, and this is where you see “SUV” or standard uptake value often on the PET scan reports.
This sensitivity is also what makes PET scans more challenging to interpret. FDG uptake is not specific to cancer. Inflammation, infection, tissue repair, and certain autoimmune or inflammatory conditions can all produce increased signal. Sarcoidosis is a classic example of a benign condition that can closely mimic malignancy on PET imaging.
Spatial resolution can affect things as well. For a lesion to be reliably detected on PET, it generally needs to be on the order of a centimeter or larger. Small lesions can appear PET-negative even when they are cancerous. A normal PET scan does not mean cancer is absent at a microscopic level. It means there is no metabolically active disease large enough to be detected.
PET scans expose patients to more radiation than CT alone and are more expensive. For that reason, they are typically reserved for situations where metabolic information is likely to change management. This is particularly true in cancers like lymphoma and sometimes melanoma, where treatment response can be better reflected by changes in activity than by changes in size, and where skin or soft tissue disease can be difficult to assess with CT alone.
Unlike CT scans, PET scans usually do not require oral or IV contrast, which some patients prefer.
MRI: high soft-tissue contrast without radiation
MRI uses magnetic fields and radiofrequency signals rather than ionizing radiation. It provides excellent soft-tissue contrast and is especially useful for imaging the brain, spinal cord, liver, pelvis, and certain musculoskeletal and soft-tissue tumors.
MRI is often chosen when anatomic detail is critical or when limiting radiation exposure over time is a priority, particularly in younger patients or those expected to undergo long-term surveillance. It can also help clarify findings seen on CT or PET.
The limitations of MRI are largely practical. Scan times are longer, and the confined space can be difficult for patients with claustrophobia. Motion sensitivity can also affect image quality. These are all factors we consider as part of the decision-making process.
Ultrasound: targeted evaluation without radiation
Ultrasound uses sound waves and involves no radiation exposure. It is commonly used for breast imaging, thyroid nodules, superficial lymph nodes, and for guiding biopsies.
Ultrasound is well suited for targeted questions, particularly in superficial structures. It is less useful for comprehensive staging or whole-body assessment and is more dependent on operator technique, which limits its role in many cancers.
How imaging decisions come together
Choosing an imaging test reflects a combination of disease biology, clinical context, and patient-specific factors. The type of cancer, typical patterns of spread, phase of care, and the specific clinical question all play a role.
Patient comfort and preferences matter. If MRI is intolerable for someone, that affects the plan. If cumulative radiation exposure is a concern, alternatives are considered when feasible. Some cancers behave in ways that make one modality far more informative than another.
There are also system-level constraints. Insurance coverage and prior authorization requirements can influence which scans are available and how often they can be performed. These limitations can be frustrating, but they are part of the reality clinicians and patients navigate together.
Questions people still ask after we talk about imaging
Why don’t all patients get routine scans after finishing cancer treatment to look for recurrence?
For some early-stage cancers, including early-stage breast cancer, routine surveillance scans have not been shown to improve outcomes. Recurrences in these settings are often first detected by symptoms, physical exams, or routine labs rather than imaging.
Regular scanning also increases false positives, anxiety, radiation exposure, and follow-up procedures without clear benefit for many patients. Imaging after treatment is usually tailored to recurrence risk, cancer biology, and whether early detection would clearly change management.
Can scans be combined to get the best information?
Sometimes. PET-CT is a common example, combining metabolic and anatomic information. In other cases, different scans are done at different times to answer different questions.
More imaging is not always better. Each test adds cost, radiation, and the chance of incidental findings that may not be clinically meaningful. The goal is the right information, not the most information.
Should I be worried about all this radiation?
Radiation exposure from medical imaging is real and cumulative. That said, the dose from any single scan is small, and modern imaging protocols are designed to minimize exposure.
In cancer care, scans are ordered because the information they provide is expected to outweigh that risk. For patients who need repeated imaging, especially younger patients, radiation exposure is actively considered when choosing modalities and spacing scans.
If MRI doesn’t use radiation, why don’t we use it more than CT?
MRI has advantages, but it also has limits. It takes longer, is more sensitive to motion, and can be difficult for people with claustrophobia. It is also not the best tool for every organ system or cancer type.
CT scans are faster, more widely available, and better suited for imaging the lungs and many abdominal structures. In many cases, CT provides exactly the information needed in a more practical way.
Why does contrast make me feel warm or like I have to pee?
This is common with iodine-based contrast used for CT scans. The sensation comes from temporary blood vessel dilation and how the contrast circulates through the body. It can feel intense but usually lasts only seconds and is harmless.
MRI contrast typically does not cause the same sensation.
If I’m allergic to shellfish, can I still get iodinated contrast?
Yes. Shellfish allergies are related to proteins in shellfish, not iodine itself. Having a shellfish allergy does not automatically increase the risk of a contrast reaction.
What matters more is whether you’ve had a prior reaction to contrast. That history should always be discussed, and there are strategies to reduce risk or choose alternative imaging when needed.
Imaging works best as part of an ongoing conversation, not something you’re left to interpret on your own.
Other questions about imaging in cancer? Post them here and I will run through as many as I can.
Did you find this article helpful? Consider upgrading to a paid subscription which helps Curative move up the “Best Sellers” list on Substack and help others find these articles more readily. Your support so appreciated.
Other articles that may be helpful along with this one:


Thank you posting this information. I was diagnosed with small cell neuroendocrine cervical ca (ki 67> 95) and am almost 5 years post treatment. My surveillance for the first two years was a CT every 3 months. Then year 3, a 4 month interval. Year 4 and 5 switching to alternating PETCT and CT every 6 months plus Natera between scans.I’m also an RN with the bulk of my career working in a busy NYC emergency department but after cancer treatment am semi-retired and have found more poignant and fulfilling work in the Radonc department (where I was treated).
I find that another important thing to mention is the importance of the interventional radiologist reading your scan. Sometimes physicians and patients can /should request a second set of eyes if they FEEL necessary.
Due to a horrific accident I have spine fusion plus other metals so MRI is out. Due to so many CT scans after trauma I developed a reaction to contrast. Recently used ultrasound for breast exam(1st one ever) and consequently needs follow up smashogram to verify findings compared to past mammo’s.
Sometimes it can be complicated..🤔